Hourly Wage for Advertising Director Salary in the United States
Hourly Wage for Advertising Director Salary
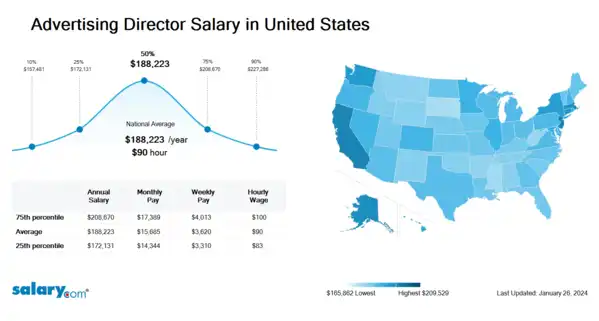
How much does an Advertising Director make hourly in the United States? The average hourly wage for an Advertising Director in the United States is $91 as of March 26, 2024, but the range typically falls between $83 and $101. Hourly rate can vary widely depending on many important factors, including education, certifications, additional skills, the number of years you have spent in your profession. With more online, real-time compensation data than any other website, Salary.com helps you determine your exact pay target.
| Percentile | Hourly Pay Rate | Location | Last Updated |
| 10th Percentile Advertising Director Salary | $76 | US | March 26, 2024 |
| 25th Percentile Advertising Director Salary | $83 | US | March 26, 2024 |
| 50th Percentile Advertising Director Salary | $91 | US | March 26, 2024 |
| 75th Percentile Advertising Director Salary | $101 | US | March 26, 2024 |
| 90th Percentile Advertising Director Salary | $110 | US | March 26, 2024 |
WORLD SYNERGY ENTERPRISES INC - Beachwood, OH
Neptune Retail Solutions - Cincinnati, OH
The Mars Agency - Cincinnati, OH
Director, Digital Marketing & Sales Enablement
Anvl - Cincinnati, OH
- View Average Salary for United States
-
Select State
-
Select City
-
Choose Similar Job
-
Pick Related Category
- View Cost of Living in Major Cities
What skills does an Advertising Director need?
Each competency has five to ten behavioral assertions that can be observed, each with a corresponding performance level (from one to five) that is required for a particular job.
Presentation: Presentation conveys information from a speaker to an audience. Presentations are typically demonstrations, introduction, lecture, or speech meant to inform, persuade, inspire, motivate, build goodwill, or present a new idea/product.
Sales Process: Designing and implementing repeatable steps that a salesperson takes to move a prospect from an early-stage lead to a closed customer.
Digital Marketing: Digital marketing is the marketing of products or services using digital technologies, mainly on the Internet, but also including mobile phones, display advertising, and any other digital medium. Digital marketing's development since the 1990s and 2000s has changed the way brands and businesses use technology for marketing. As digital platforms are increasingly incorporated into marketing plans and everyday life, and as people use digital devices instead of visiting physical shops, digital marketing campaigns are becoming more prevalent and efficient. Digital marketing methods such as search engine optimization (SEO), search engine marketing (SEM), content marketing, influencer marketing, content automation, campaign marketing, data-driven marketing, e-commerce marketing, social media marketing, social media optimization, e-mail direct marketing, Display advertising, e–books, and optical disks and games are becoming more common in our advancing technology. In fact, digital marketing now extends to non-Internet channels that provide digital media, such as mobile phones (SMS and MMS), callback, and on-hold mobile ring tones. In essence, this extension to non-Internet channels helps to differentiate digital marketing from online marketing, another catch-all term for the marketing methods mentioned above, which strictly occur online.
Job Description for Advertising Director
Advertising Director directs and develops the strategy, plans, and tactics required to create advertising concepts and campaigns that deliver results and meet organizational objectives. Oversees the budget, expenditures, and resource allocation for all advertising campaigns. Being an Advertising Director identifies media channels and directs the placement of all advertisements and the development of promotional materials. Ensures consistent and effective branding and messaging in all forms of advertisements. Additionally, Advertising Director uses metrics and data analytics to measure campaign effectiveness and assess return on investment. Fosters innovation of new advertising methods and initiatives using advertising industry experience and competitive intelligence. Requires a bachelor's degree in marketing, advertising or equivalent. Typically reports to top management. The Advertising Director manages a departmental sub-function within a broader departmental function. Creates functional strategies and specific objectives for the sub-function and develops budgets/policies/procedures to support the functional infrastructure. To be an Advertising Director typically requires 5+ years of managerial experience. Deep knowledge of the managed sub-function and solid knowledge of the overall departmental function. (Copyright 2024 Salary.com)... View full job description
See user submitted job responsibilities for Advertising Director.
Search Job Openings
Salary.com job board provides millions of Advertising Director information for you to search for. Click on search button below to see Advertising Director job openings or enter a new job title here.
Career Path for Advertising Director
A career path is a sequence of jobs that leads to your short- and long-term career goals. Some follow a linear career path within one field, while others change fields periodically to achieve career or personal goals.
For Advertising Director, the first career path typically progresses to Top Advertising Executive.
What does an Advertising Director do?
Are you an HR manager or compensation specialist?
Salary.com's CompAnalyst platform offers:
- Detailed skills and competency reports for specific positions
- Job and employee pricing reports
- Compensation data tools, salary structures, surveys and benchmarks.

Advertising Director Pay Difference by Location
Advertising Director salary varies from city to city. Compared with national average salary of Advertising Director, the highest Advertising Director salary is in San Francisco, CA, where the Advertising Director salary is 25.0% above. The lowest Advertising Director salary is in Miami, FL, where the Advertising Director salary is 3.5% lower than national average salary.
| City, State | Compared to national average |
|---|---|
| City, State San Francisco, CA |
Compared to national average
|
| City, State Washington, DC |
Compared to national average
|
| City, State Miami, FL |
Compared to national average
|
| City, State Chicago, IL |
Compared to national average
|
| City, State Boston, MA |
Compared to national average
|
| City, State New York, NY |
Compared to national average
|
| City, State Dallas, TX |
Compared to national average
|
Similar Jobs to Advertising Director

| Job Title | Experience | EDUCATION | Salary Compared to This Job |
|---|---|---|---|
| Job Title Advertising Coordinator I | Experience 0 - 2 | EducationBachelors | Salary Compared to This Job |
| Job Title Advertising Coordinator II | Experience 2 - 4 | EducationBachelors | Salary Compared to This Job |
| Job Title Advertising Coordinator III | Experience 4 - 7 | EducationBachelors | Salary Compared to This Job |
| Job Title Advertising Coordinator IV | Experience 7 + | EducationBachelors | Salary Compared to This Job |
| Job Title Advertising Coordinator V | Experience 10 + | EducationBachelors | Salary Compared to This Job |
Level of Education for Advertising Director
Jobs with different levels of education may pay very differently. Check the Advertising Director salary of your education level.
Advertising Director Salary by Global Country
Advertising Director salary varies from country to country. There are several factors that mainly impact the Advertising Director salary, including cost of living, economic conditions, market rates and legal differences. Click below to Advertising Director salary of the other country.
Advertising Director Salary by State
Geographic variations impact Advertising Director salary levels, due to various factors, such as cost of living, industries, market demand and company budgets. Click below to see pay differences between states.
Browse All Advertising Jobs by Salary Level
Browse Related Job Categories With Advertising Director
A job category is a classification or grouping of job positions that share similar characteristics, functions, or industries. Advertising Director salary varies from category to category. Click below to see Advertising Director salary in different categories.
Take just three simple steps below to generate your own personalized salary report
Understand the total hourly compensation opportunity for an Advertising Director, hourly wage plus other pay elements
Average Hourly Wage
Core compensation
Average Total Hourly Cash Compensation
Includes base and short-term incentives
View the Cost of Living in Major Cities
Skills associated with Advertising Director: Marketing Communications, Project Planning, Customer Insights, Media Planning ...More
Recently searched related titles: Director Of Promotions
Jobs with a similar salary range to Advertising Director : Advertising Sales Consultant, Advertising Sales Manager, Marketing Director, Strategic Communications
Salary estimation for Advertising Director at companies like : Hill Center Lansing School District, Healthcare Clinics, Mayuri Group International Corp
Jobs with a similar salary range to Advertising Director : Campaign Executive